How GCMI supported Georgia Tech Spring 2022 Capstone Team UCOLLECT’s Need for Verification Testing on the pathway to commercialization.
“More than 6 in every 10 women will, at some point in their life, be unable to toilet. For these women, the best options include the Foley catheter, a device that is inserted up the urethra and can cause infection and damage if not managed properly. Currently, there is a lack of external urinary collection devices for women with incontinence that keep them dry and can be used in both sitting position and when they move around.”
So states the Georgia Tech Spring 2022 BME Capstone Team UCOLLECT.
Key stakeholders include not only the patients themselves, but also their care team, including nurses, nursing assistants, and their doctors, who prescribe solutions to incontinence. The global incontinence market size is about 10.12 billion a year, as 80 percent of patients are women. SAM for this product would be around 8 billion and SOM focusing only on the market in the United States would be about 506 million.
“Let’s face it, no one is interested in wearing or changing adult diapers. And use of catheters for the job over extended periods of time invites complications that absolutely should be avoidable,” Team UCollect’s Josie Benner (Georgia Tech BME Class of 2022) told us.

Dr. Janelle Foote. Image credit: Emory Healthcare
“The failure of existing products on the market to successfully accomplish the needs of patients and clinicians made this a problem we addressed with enthusiasm and with the support and encouragement of Dr. Emily Blum and Dr. Jenelle Foote especially with regard to patient, user and clinical needs.”
The solution also needed to prevent backflow and urine contact with skin, a user and patient need. If a device for this purpose and need is gravity fed, fluid is going to want to go ‘down’ whatever direction down is.
GCMI has proudly supported select Capstone teams for years with our medical device technology design and development capabilities, facilities, staff and affiliated resources including preclinical testing and bioskills training assets.
The right resources for the right team in the right place at the right time
“When Evan Goldberg visited with the Capstone teams and made us aware of the resources available to us at GCMI, I immediately called Rhonda Lynch,” Josie said. Rhonda is program manager for training and education in GCMI’s preclinical arm also known as T3 Labs. Evan is director of scientific affairs for GCMI.
“When she told us they have a cadaver that voids and we could use for our design verification testing, we jumped on the opportunity. The absence of suitable, functional and anatomically accurate models at reasonable prices for our project made this contribution invaluable to our team’s work and results.
“Using humans for our testing needs was not a viable option due to the absence of controls for volume.
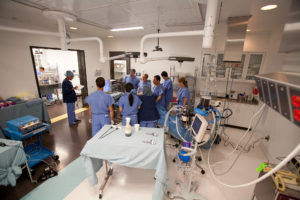 “We had the access to the same facilities, staff and resources, which may have included the same voiding cadaveric abdomen, a major medical device company used for its technology, which is a phenomenal asset to academic teams and projects at our early phase of innovation. Rhonda and her team were very tuned in and highly focused on our work together, and for all this the team is thankful.”
“We had the access to the same facilities, staff and resources, which may have included the same voiding cadaveric abdomen, a major medical device company used for its technology, which is a phenomenal asset to academic teams and projects at our early phase of innovation. Rhonda and her team were very tuned in and highly focused on our work together, and for all this the team is thankful.”
The team’s cadaveric verification testing informed materials and design changes ultimately demonstrating the product had less than 1 percent leakage, a remarkable improvement from the original, under 15 percent, goal.

The result
The team’s solution, informed by the verification testing supported by GCMI, “is an interlabial external urinary collection device designed for women, and comes in two main parts: the collection cup, and the attachment mechanism. [It is] designed for size inclusivity with a variety of attachment methods suitable for women of all ages.
“The collection cup covers the female vulva area around [the] urethra opening for maximal urine collection. It prevents the backflow of urine with a catch canal around the cup inside to minimize skin irritation from residual urine. The device can be kept attached to the body through a number of different mechanisms, including an intravaginal method, modeled after devices that are already on the market for intravaginal use, or with our specially modified underwear. The device is compatible with current urine collection bags, which can be attached to the leg or wheelchair of the patient for urine drainage. The device is washable and reusable, which makes it much more eco-friendly than single use catheters or diapers.”
From a regulatory perspective, because the device has two individual components there are two options. An “underwear modified” solution would fall into the FDA’s Class I (external to body with very little risk of harm) device classification. The second option, or “attachment method” via the inter-vaginal cup would qualify as a Class II product or technology.
Because the team has scattered into their next opportunities in disparate locations, they are in the process of filing for a provisional patent in the Class II category that will protect the intellectual property for the time being and have handed off the work to Dr. Foote to carry forward. Because of the number and size of the competitors in the market, the team believes there is high potential for an industry leading medical device company to license the patent, refine the design where needed to successfully commercialize the product.
What did the team learn from activities completed at GCMI / T3 Labs?
- Play to the team’s strengths.
“Everything is not going to be ‘your thing.’ One team member had awesome graphic design skills. Another had ‘few’ of Yilin’s design skills, but Yuyan Cai was completely comfortable and proficient in the cadaveric testing work. Play to each others’ strengths and support them with your own.”
- Prepare.
“There is always a good chance some element of a project will fail. Think about the range of possibilities, prepare for eventualities and bring backups. In our case that meant multiple prototypes in multiple sizes each.”
Currently still a student at Georgia Tech until the end of July, Josie is also working at Merck Animal Health as an External Manufacturing Quality Co-Op. We thank Ms. Benner for helping us share the team’s story and wishes each member continued success in their professional journeys.
About GCMI
GCMI’s mission is to bring new medical technologies to market that improve quality-based outcomes and delivery of care for patients worldwide. This certainly includes supporting development of medical technologies born in our backyard from faculty, researchers and students with whom we share our Georgia Tech institutional affiliation. We congratulate the Spring 2022 teams and eagerly anticipate what the Fall 2022 teams have to show us later this year.