What you need, when you need it
For our orthopedic medical device testing and training sponsors, our GLP and AAALAC accredited preclinical CRO employs the most appropriate, efficient model and all equipment needed.
- Digital x-ray
- Micro-CT
- MRI and CT
- Surgical instrumentation
- Qualified plastic histology labs
- Qualified mechanical testing labs
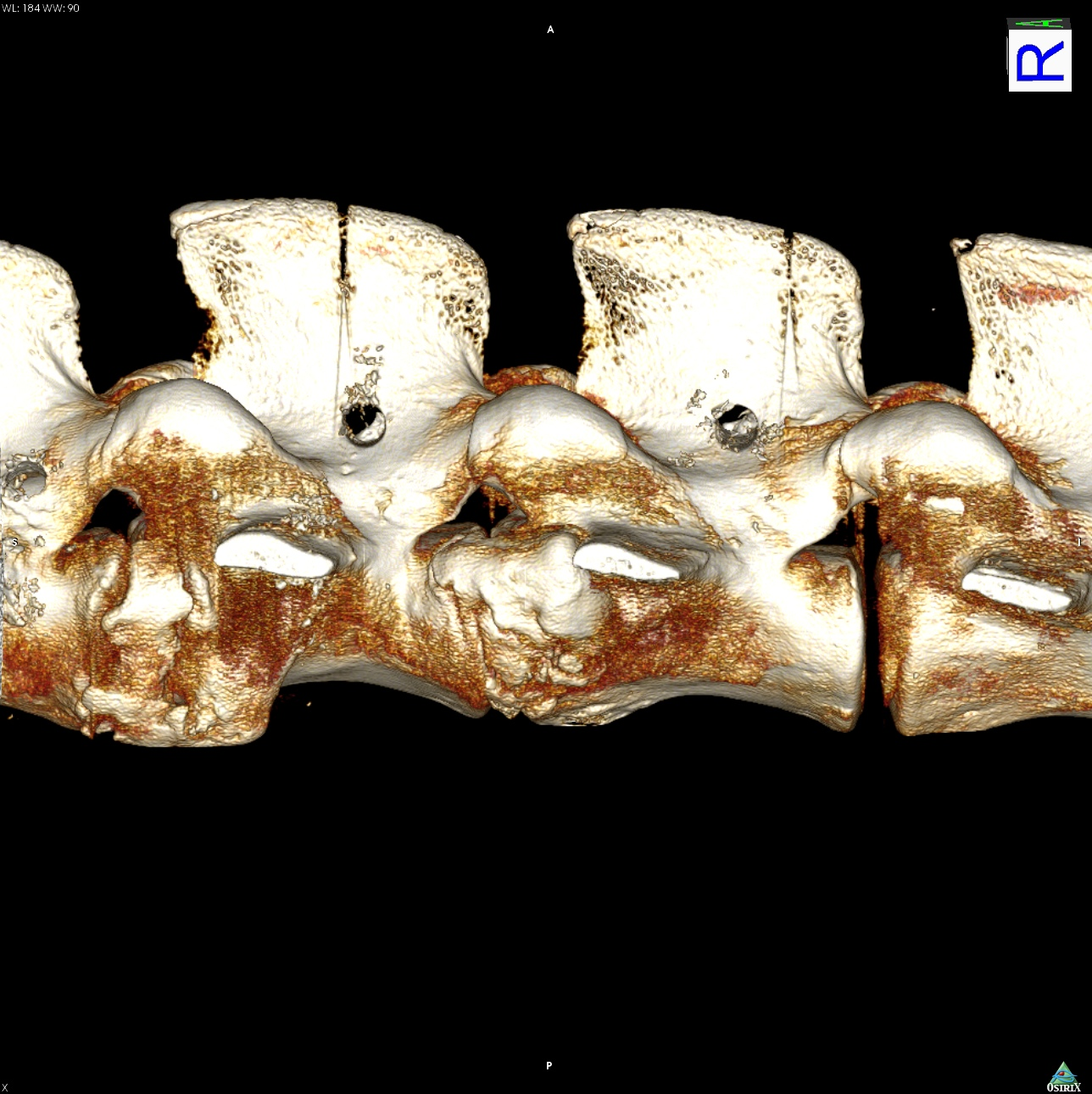
One streamlined preclinical CRO engagement for orthopedic device testing
Our relationships with experienced, practicing orthopedic surgeons can expedite your orthopedic device testing program as well. This eliminates the need for orthopedic manufacturers to engage in additional contracts with other physicians, CRO or labs. It also brings deep knowledge creating seamless simplicity and economy via one preclinical CRO engagement. Examples include:
- Emory Orthopedics and Spine Center
- Georgia Institute of Technology Petit Institute for Bioengineering and Bioscience
- GCMI (the Global Center for Medical Innovation, experts in rapid prototyping of new medical products) bring deep knowledge creating seamless simplicity and economy via one preclinical CRO engagement.
Experience
Our GLP preclinical program for orthopedic devices includes extensive experience in:
- Tendon repair/replacement
- Bone segmentation/fracture healing
- Osseointegration
- Spine fusion
- Infection
- Degenerative disc disease
- Prosthetics/amputation
- Osteochondral defect
Our orthopedic device testing and training program leaders are scientists as well
Our orthopedic device testing and training program leaders have extensive bioengineering and veterinary backgrounds enabling them to intimately understand your orthopedic device’s intentions and prospective value.
Our preclinical team includes multiple biologists, PhDs, surgical technicians and DVMs with more than 50 years of collective experience in the science. The vast majority of our reports submitted to the FDA return with zero questions asked.